On October 13,2021, the Center for Drug Evaluation (CDE) of has granted BeBetter Med’s BEBT-908 "Breakthrough Therapy " designation, which is used for the treatment of patients after at least two systemic treatments of relapsed or refractory diffuse large B-cell lymphoma (r/r DLBCL).
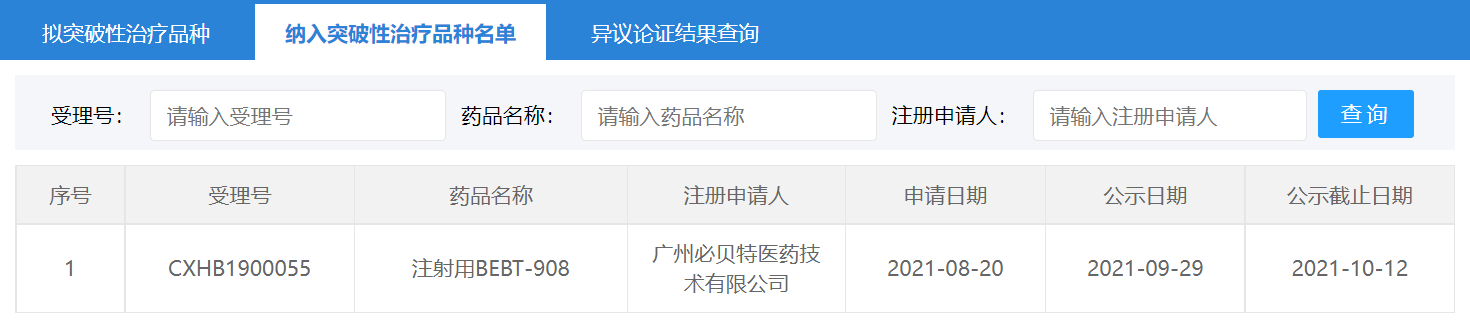
BEBT-908 is a first-in-class PI3K/HDAC dual-target anti-tumor drug invented by Dr. Qian Changgeng and Dr. Cai Xiong, founders of BeBetter Med Inc. It can selectively inhibit the core target that is crucial to tumor survival. It can synergistically destroy tumor signaling pathways, and have a strong inhibitory effect on a variety of hematological tumors and solid tumors. The preclinical study results were published in the well-known oncology journals Clinical Cancer Research (2012) and Cancer Research (2021). The Phase IIa clinical study of BEBT-908 in the treatment of r/r DLBCL was completed in the second quarter of this year, and a pivotal Phase II clinical trial is currently underway.
DLBCL is the most common aggressive non-Hodgkin lymphoma. It is estimated that the number of new DLBCL cases in China is about 34,500 per year, of which 35-40% of DLBCL patients are initially refractory or relapsed after initial treatment. r/r DLBCL after two or more systemic treatments is highly invasive, disease progresses rapidly, and has short survival periods, which present important unmet clinical needs. The clinical results of BEBT-908 have shown excellent anti-tumor efficacy and good safety, which earned Breakthrough Therapy designation. This will greatly accelerate its launch onto the market, which will fill the blank for the currently absent effective r/r DLBCL-targeted drug therapy in China, and solve the urgent needs for the treatment of r/r DLBCL patients.
In addition to the pivotal Phase II clinical trial for the treatment of r/r DLBCL indication, other ongoing clinical trials of BEBT-908 include: (1) Phase II clinical trials of BEBT-908 in the treatment of relapsed and refractory T-cell lymphoma; (2) Phase II clinical trials of BEBT-908 in the treatment of relapsed and refractory follicular lymphoma, marginal zone lymphoma and lymphocytic leukocytes; (3) BEBT-908 monotherapy in the treatment of PIK3CA mutant advanced solid tumors, combined with Fulvestrant in the treatment of ER+/HER2- advanced breast cancer, three Phase Ib/II clinical trials combining PD1/PD-L1 monoclonal antibody in the treatment of advanced solid tumors; (4) Phase Ib/II clinical trial of BEBT-908 combined with BEBT-109 in the treatment of advanced NSCLC resistant to third-generation EGFR inhibitor therapy; (5) Phase Ib/II clinical trial of BEBT-908 combined with BEBT-209 in the treatment of ER+/HER2- advanced breast cancer resistant to endocrine therapy and CDK4/6 inhibitor therapy.
Related News
- BEBT-503 Passes Phase I Clinical Trial in Australia, Marking a Milestone in BeBe...
- BeBetter Med’s Pan-mutant EGFR Inhibitor BEBT-109 Phase III Clinical Trial Appl...
- BeBetter Med’s First-in-Class New Drug BEBT-908 NDA Accepted
- BeBetter Med Inc. successfully holds the sponsors meeting & the first general me...
- BeBetter Med’s BEBT-209 Approved for Phase III Clinical Trials
- First-In-Class Drug RGRN-305 (BEBT-305) Clinical Proof-of-Concept Study Results ...
- First-In-Class Drug BEBT-908 Research Results of BEBT-908 Published on CANCER RE...
- BeBetter Med’s BEBT-908 granted Breakthrough Therapy designation


